Archive for May, 2011
POZEN’ a threat to outdated pharma marketing
Thursday, May 26, 2011, 3:42 pm No Comments | Post a CommentOriginally published on TheRTP Blog, 5/25/11. Watch The RTP’s exclusive sit-down interview with Liz!
For 25 years, Liz Cermak worked for Johnson & Johnson, one the biggest names in the pharmaceutical business. Now, she works for one of the smallest. And she says in terms of marketing, it’s giving the Goliaths a run for their money.
Cermak is the executive vice president of POZEN, Inc., a Chapel Hill-based pharma developer/manufacturer with no more than 30 employees. Since coming onboard in 2009, she has overseen the company attain FDA approval on two authentic combination drugs: Treximet and VIMOVO—for migraines and osteoarthritis, respectively. No small feat, even by J&J standards.
But what Cermak is most excited about is POZEN’s fresh and unique approach to pharmaceutical marketing.
Instead of sending sales representatives to hospitals and doctors’ offices to promote their products, Cermak and her team pitch most their medicines online.
“The reality is that the current sales rep model of traditional pharma is obsolete,” Cermak said to a packed house at the Marketing Mondays series held at The Research Triangle Park HQ earlier this week. “Eighty-six percent of US doctors go online for product info now, and 82 percent are on smart phones.”
In-person sales pitching can be inefficient, she said, because all health care workers are overbooked and overbusy, and representatives must endure a costly wait just to get two minutes in with the doctor.
Two minutes. That’s the average rep-doc face time. But online, the average time spent by a physician on a single ePromotion activity is eighteen minutes.
Cermak has three rules for digital pharma marketing:
1. Develop products that deliver real value to customers.
Be relevant and learn from your customers. Understand their needs and study their e-behavior. Most pharmaceutical companies need to broaden their apertures here, she said.
2. Make them affordable and accessible.
POZEN recognizes the strains today’s pharmaceutical pricing puts on doctors and patients today alike. As should go without saying, costs must be kept low to compete and to demonstrate a respect for your consumers.
3. Engage with customers in a meaningful but highly efficient way.
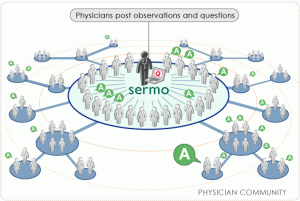
This means using social media and online public networks like Facebook and LinkedIn, but also more exclusive, MD-only communities like Sermo or CogNet. Use push and pull marketing tactics; see what works and what doesn’t.
Cermak calls this “Pharma 3.0”.
“The change isn’t coming,” she said. “It’s here.”
We’ve seen this before with other industries, as well. Amazon now sells more books for the Kindle than it does in print, and Netflix’s superior, customer-based business model has Blockbusters closing up shop around the country. The global economy is now decidedly digitalized and will only continue to shift that way.
Now, as POZEN enters the final testing and approval phase for its latest development, an ulcer-reducing aspirin compound dubbed PA32540, a viral campaign is already underway to spread the word.
Cermak stressed there is still utility in face-to-face interaction, though. Sending sales reps is important to explaining drug principles to doctors, learning about clinic demographics, and building a personal rapport with primary care physicians. However, there are not enough reps to go around as it is now, and focusing sales online will drastically cut down their jampacked schedules.
The biggest advice Cermak has for pharma companies looking to try this new approach is to not be afraid to experiment. To take risks. And to lose.
“Be ready to try and fail,” she said. “Absolute ROI of a given digital initiative cannot be accomplished with a high degree of certainty.”
No one expected it to work out for POZEN. But no one expected 30 people from Chapel Hill to get two drugs FDA-approved in two years, either.
Webby wins highlight gaming as lifestyle
Monday, May 23, 2011, 7:30 pm No Comments | Post a Comment Russ Pitts finds it hard to remember a time when he wasn’t playing video games.
Russ Pitts finds it hard to remember a time when he wasn’t playing video games.
From the Magnavox Odyssey to the Fairchild Channel F, Pitts embraced gaming as a kid in the 70s — and he hasn’t let go since.
“My whole childhood was filtered through this lens of games,” Pitts said.
That’s why it’s not a big surprise that Pitts is now at the helm of The Escapist, an online gaming magazine based in Durham. Now approaching its sixth-year anniversary, The Escapist competes for gamers’ attention with heavy hitters like IGN and Gamespot. Both sites — and others like them — are filled with reviews, walkthroughs and trailers to guide gaming consumers.
“Our challenge has been trying to find our footing as we grow to the size of these gargantuan companies,” Pitts said. “Our success has put us toe-to-toe with some of these giants.”
But The Escapist has a different strategy. Read more…
Does the FDA ensure that nanotech medicines are safe?
Sunday, May 22, 2011, 11:35 pm No Comments | Post a CommentNanotechnology promises to make medicines more effective, reduce side effects and allow earlier diagnoses of diseases. But nanotechnology also raises multiple questions most of which, for now, remain unanswered.
When the N.C. Center of Innovation for Nanobiotechnology, also known as COIN, brought together a panel of regulatory experts in Research Triangle Park, several entrepreneurs and developers sought guidance on how to develop safe and effective nanotechnology products, especially from panel member Katherine Tyner, a chemist who heads a small nanotechnology research group at the Food and Drug Administration’s Center for Drug Evaluation and Research.
Understanding FDA requirements is crucial to get new drugs and medical devices approved for sale, but the FDA doesn’t have specific guidelines for nanotechnology. The agency doesn’t even have an official definition for nanotechnology, Tyner said. To assess safety risks of a nanotechnology product and whether it works, regulators apply existing guidelines for products without nanotechnology on a case-by-case basis.
It’s a system that works, said Lawrence Tamarkin, chief executive of CytImmune, a Rockville, Md.-based nanomedicine company, who also sat on the COIN panel on May 17.
Tamarkin suggested the great variety in nanotechnologies doesn’t lend itself to common guidelines. “It’s better the way we do it now,” he said.
Tyner, who researches drug safety but doesn’t review requests to approve nanotechnology products for sale, listed about two dozen medicines, cosmetic products and contrast agents for diagnostic tools that use nanotechnology and have passed muster with the FDA in the past 15 years.
But the list didn’t go over well with Jamie Oliver, a pharmacist and the chief executive of Peptagen, a Raleigh startup working on a nasal spray that delivers nanoparticle vaccines. Oliver, who sat in the audience during the COIN panel, shook his head when asked about Tyner’s FDA-approved nanomedicines list.
“No true nanoparticle [drug] is on the market,” he said.
Nanotechnology has been used for years in material science, electronics and cosmetic products, according to the National Nanotechnology Initiative. Nanoparticles make tennis rackets stronger, fabric wrinkle-free and camera lenses self-cleaning. Engineers have shrunk resistors to nanoscale to make computers smaller and more powerful.
Nanotechnology is also used to make batteries more efficient and to power mobile devices. A nanofabric paper towel cleans up 20 percent more spilled crude oil in an environmental cleanup. Many sunscreens, shampoos and lotions contain nanoparticles.
Though the applications are diverse, they all have one thing in common: The technology is generally only called nano when it measures between 1 nanometer and 100 nanometer.
One nanometer is about 100,000 times smaller than a hair is thick. Hemoglobin, the oxygen-transporting protein in red blood cells, fits the official nanoscale, so does HIV, the virus that causes AIDS, and the width of a cell membrane. Single-walled carbon nanotubes and nanosilver are also true nanoparticles.
XinRay Systems, a medical device startup in RTP, uses carbon nanotubes-studded film to develop X-ray machines that are faster and more accurate. (More about XinRay Systems here.)
Nanosilver is a natural germ killer and is used in anything from pacifiers to sunscreen.
Lacking a definition for nanotechnology, the FDA considers even structures ten times larger than 100 nanometer as nanoparticles, such as liposomes, man-made vessels that are filled with drugs. Liposomes tend to measure more than 1,000 nanometers.
But size is crucial to nanotechnology. Richard Feynman, the father of nanotechnology, pointed that out in 1959. In “There’s Plenty of Room at the Bottom,” Feynman wrote that in a very, very small world, “we are working with different laws, …, we have new kinds of forces and new kinds of possibilities, new kinds of effects.”
Half a century later, researchers have just begun to look into some of those forces and effects that only occur on a true nanoscale.
Case in point: nanosilver.
Products that use nanosilver as an antibacterial contain silver particles that are 10 nanometers and 50 nanometers, but also silver particles that are much larger. Nanosilver was considered safe enough to be used in baby products, such as blankets, pacifiers and bottles.
But a 2008 study by researchers from the University of Connecticut’s Center for Environmental Sciences and Engineering raised concerns that loose nanosilver could compromise the human immune system. The smaller particles had that effect but not the larger ones, according to a ScienceNews report.
Since then, the Environmental Protection Agency has started to study the effects loose nanosilver has on the environment. (More on a petition to have nanosilver declared a pesticide here.)
So without a definition for nanotechnology in place, will the FDA catch potential safety risks that are specific to particle size?
Tyner presented an FDA that is very confident in the way it regulates nanotechnology products. But research projects under way at the FDA suggest regulators worry about missing clues for potential risk factors. Researchers at the agency are working on improved methods and tools to detect and measure nanomaterials and assess their safety and efficacy. And they’re developing class-based approaches to risk assessment of nanotechnology products.
NC history of flight extends to space
Thursday, May 19, 2011, 9:26 pm No Comments | Post a CommentNASA’s interest in North Carolina goes back to the 1960s, when U.S. astronauts came to the University of North Carolina at Chapel Hill to learn at the Morehead Planetarium how to navigate in space by looking at stars, according to a N.C. Museum of History report. In the past 25 years, a handful of North Carolinians flew on space shuttle missions, including Beaufort native Michael Smith, who died in 1986 when the Challenger exploded shortly after liftoff.
To prepare for and support NASA missions, researchers at N.C. State University have studied how to more precisely land a vessel on Mars and how to grow plants in a spacecraft operating in zero gravity.
But NASA’s involvement in North Carolina goes further. Since 1991, teachers and students at North Carolina universities, community colleges and public schools have received about $15 million to study science, technology, engineering and mathematics, to research issues related to space missions and to work in companies contracting with NASA.
Funding for the research grants, scholarships and summer internships has been provided through the N.C. space grant, a program administered at NCSU.
“The goal is to keep the pipeline filled for NASA,” Christopher Brown, director of the N.C. space grant, told members of the Triangle Area Research Directors Council who gathered this week at Research Triangle Park headquarters.
But “this isn’t just rockets and aerospace,” Brown said. Space grant projects in North Carolina include satellite tracking of red wolves and the development of an undergraduate robotics course at Duke University.
As a professor of plant biology at NCSU, Brown teaches a space biology class and some of his plant experiments will travel to the international space station on the last shuttle flight scheduled to take off in August.
NASA funds the N.C. space grant and similar programs in all other states through its annual budget.
In the past five years, the total for these grants has increased from about $30 million to $45.6 million. North Carolina’s portion is about $800,000 annually, Brown said. That includes a state match. The match used to be about $200,000 per year, but budget cuts have reduced it to $180,000.
Federal cuts are also looming, but Brown said he didn’t think the program would be eliminated, because every Congressional district receives money. For fiscal year 2012, which starts Oct. 1, President Obama’s budget request for the space grant program is $26.5 million, according to NASA budget information. That’s a reduction of more than 40 percent.
In the past, N.C. space grant money has supported research of young university faculty, helped develop new college courses and paid for professional development of K-12 teachers, provided scholarships and summer internships for graduate and undergraduate students and students at community colleges.
Thirteen university campuses across North Carolina are affiliated with the N.C. space grant, from UNC Asheville to Elizabeth City State University.
Gears of War 3 beta wraps up, but more for fans
Tuesday, May 17, 2011, 8:24 am No Comments | Post a Comment
Augustus "Cole Train" Cole blasts an enemy locust soldier to save wounded teammate Anya Stroud. Gears of War 3 was available in private beta until Sunday. | Photo courtesy of Epic Games
Cary-based Epic Games had more announcements this week for fans despondent over the end of the Gears of War 3 multiplayer beta.
The special preview of the anticipated title, which allowed a select few to battle each other to the death on new maps, ended Sunday night. While the full version of the game isn’t due out on store shelves until Sept. 20, Epic dropped another morsel in the lap of its fan base Monday: details about the coveted limited editions.
On top of the regular game, which will retail for $59.99, the special editions will include premium downloadable content and collectables — all available only in limited quantities. Here are the details, per Epic’s press release:
Epic Edition - $149.99
• A limited edition statue of main character Marcus Fenix, designed by Epic Games Art Director Chris Perna and crafted by the artisans at Triforce.
• The “Art and Design of Gears of War” by Tom Bissell, author of Extra Lives: Why Video Games Matter. The book is a behind-the-scenes glimpse into the game, with 96 pages of interviews, photos, concept artwork and more.
• Infected Omen Weapon Pack – custom multiplayer skins for the game’s five starting weapons; Lancer, Retro Lancer, Hammerburst, Sawed-off Shotgun and Gnasher Shotgun.
• Everything in the limited edition.
Limited Edition - $79.99
• An Octus Award Box with Octus Service Medal. A 1:1 match in size and weight, this zinc-alloy cog-shaped medal replicates the award given to Adam Fenix, father of the game’s main character, in recognition of his work on the Hammer of Dawn, a devastating weapon in the war against the Locust.
• Exclusive unlockable Adam Fenix Multiplayer character – Each Octus Service Medal replica is engraved with a unique Xbox LIVE code that is the only way to unlock Adam Fenix in multiplayer.
• A fabric Coalition of Ordered Governments (COG) Flag
• The personal effects of Adam Fenix, including his “Last Will and Testament,” the initial Hammer of Dawn schematic and other Fenix family mementos.
But Epic’s not likely done hyping the crown jewel of its trilogy, the first two parts of which sold more than 12 million copies since the saga began in 2006. Franchise Executive Producer Rod Fergusson announced on Twitter last week that if Gears of War 3 wins IGN’s Most Anticipated Game Award, they’ll run a week-long event granting players 30 times the points in the Gears of War 2 multiplayer during the E3 Conference June 6.
Can’t wait until then? Then satisfy your bloodlust with this gruesome execution montage, from IGN (warning: graphic). Cheers!
North Carolina adds another renewable to its energy sources
Tuesday, May 3, 2011, 10:56 pm No Comments | Post a CommentNorth Carolina is on the way to become a state with one of the largest photovoltaic solar farms and one of the largest wind farms in the U.S.
The N.C. Utilities Commission Tuesday approved plans of a Spanish company to build up to 150 turbines, each about 400 feet tall, near Elizabeth City in the northeastern corner of the state. If the $600 million project gets the necessary federal, state and local permits, it will be another large source of renewable energy that is produced in North Carolina.
About 270 miles west of Elizabeth City, near High Point, construction of the final phase of a $173 million solar farm with 63,000 photovoltaic panels is under way on about 200 acres.
The electricity the two projects are expected to generate - enough to provide power to about 62,000 homes per year - would become part of the energy blend that residential, commercial and industrial consumers in the state already receive from the power grid. The utilities commission has been goosing North Carolina power companies and cooperatives for three years to add energy generated from renewables to the mix.
This regulatory pressure and industry incentives are key to successfully reduce America’s dependence on oil and lower the amount of harmful greenhouse gas emissions, Steve Kalland, executive director of the N.C. Solar Center, said during a recent meeting of the Triangle Area Research Directors Council in Research Triangle Park.
“It’s not a technology question anymore,” Kalland said. “Financing and regulatory are the two biggest barriers to move technologies forward.”
Energy from renewables is still more expensive than energy from traditional sources, such as oil and coal, Kalland said, but oil prices are going up and the price for green technology is coming down. “The trendlines say time is working in our favor.”
For the second time in three years, crude oil prices are above $100 a barrel and gas prices at the pump are closing in on $4 per gallon. Meanwhile, the cost to get solar panels installed in North Carolina has dropped 49 percent since 2007.
“Everytime oil goes up, we get a policy opportunity,” Kalland said. What he means by that is legislation that supports renewable energies, particularly federal legislation that deals with the differences in regulations from state to state. “Fifty states have 50 regulatory commissions, it’s something that cries out for federal intervention,” he said. But an array of special interests have so far foiled attempts to get anything done nationally.
The North Carolina legislature has done more for renewable energy supporters.
In 2007, state lawmakers established renewable portfolio standards that the utilities commission tracks by making power suppliers file compliance reports. The standards say that by 2021, 12.5 percent of the energy that investor-owned utilities like Duke Energy supply must be generated from renewable sources. Solar, wind, biomass, tidal energy, landfill gas, swine and poultry waste all qualify and consumers must pay for part of the costs.
North Carolina is one of 32 states with such standards, according to information collected by the U.S. Department of Energy.
For several years, the state has also offered a 35 percent renewable energy investment tax credit as an incentive to install solar, wind, geothermal and other renewable energy technology. Last year, the legislature added a tax credit for businesses and homeowners who install combined heat-and-power systems. CHP systems are up to twice as efficient than traditional heating and cooling systems.
If 20 percent of U.S. households installed CHP systems by 2030, the amount of energy consumed by U.S. households would be cut in half, the U.S. Department of Energy estimated.
The carrot-and-stick approach has boosted the number of solar water heating installations and photovoltaic installations in North Carolina, ranking the state in the top 10 nationwide. In 2010, more than 100 solar energy companies operated in the state, employing more than 1,500, according to a report by the N.C. Solar Center.
The N.C. Sustainable Energy Association estimated that last year about 12,500 job in North Carolina were green.
SunEdison completed construction of the photovoltaic farm near High Point in January. Duke Energy has a 20-year contract to buy all of the power generated by the farm - about 17 megawatt, or enough to supply 2,600 homes per year.
The wind farm that the U.S. subsidiary of the Spanish Iberdrola Renewables wants to build on about 20,000 acres in northeastern North Carolina is projected to produce up to 300 megawatt, or enough to supply 60,000 homes per year. The wind farm could start operations as soon as January 2013.
There’s potential for more to come off the coast.
A 9-month feasibility study that the University of North Carolina published in 2009 recommended that North Carolina pursue wind energy production aggressively. The study looked at locations offshore and in the Pamlico and Albermarle sounds and found 2,800 square miles within 50 miles of the coastline particularly well suited and worthy of further investigation.
Hurricanes are a threat to offshore wind farms, Kalland acknowledged during his TARDC talk. But insurance companies have no problem insuring the turbines.
Eisai’s RTP plant aims to supply the world
Sunday, May 1, 2011, 9:36 pm No Comments | Post a CommentEisai’s new production plant in Research Triangle Park is fully equipped to make Halaven, a new treatment for advanced breast cancer, but the 66,700-square-foot building is still empty of people except for the occasional employee mopping floors. It will stay that way until the Food and Drug Administration inspects the plant and clears it for production.
The FDA approval, which Stephen Errico, director of Eisai’s parenteral operations, expects in September, will usher in a new era for Eisai and its main U.S. manufacturing site in RTP.
The Japanese drugmaker is switching its attention to injectable drugs after focusing on pills for many years. Injectable cancer treatments such as Halaven are on top of Eisai’s priority list and no matter whether these cancer treatments will come out of Eisai’s own research and development labs or the labs of partners, the new RTP plant will make and package them and ship them, first to the U.S. market and later the rest of the world.
Plans to seek regulatory approval for the RTP plant to produce for the European market are next.
“This facility is very unique and important to Eisai,” Errico said during a recent tour of the plant.
For the past 13 years, Eisai has made and packaged pills in RTP - Aricept, the leading Alzheimer’s treatment, and Aciphex, an acid reflux treatment. Aricept, Eisai’s biggest seller, generated about $2 billion in annual sales in the U.S. But in November, Aricept lost patent protection and Eisai expects to lose about 60 percent of its blockbuster’s sales to cheaper generic competitors over the next two years. In March, Eisai cut 70 jobs at its RTP operations, all of them in the pill part of its business.
Future growth sees the company in the injectables and oncology market.
So do most pharmaceutical companies.
Cancer is the second most common cause of death in the U.S., according to the American Cancer Society. In 2010, more than 500,000 Americans died from the disease and more than 1.5 million Americans were newly diagnosed with cancer. With more than 200,000 new cases every year, breast cancer is the most common cancer in women.
The active ingredient in Havalen is eribulin mesylate, a synthetic version of a chemical made by a black sea sponge. It was first isolated in 1985 by a Japanese scientist and has shown to prevent cell division. Eisai found it screening chemicals made by plants and animals living under water and in the tropical rain forest.

Eisai's operations in RTP include a new plant to make injectable oncology drugs. The new plant, in the foreground, could be expanded where the parking lot is now.
Eisai makes the eribulin mesylate in Japan. The active ingredient arrives in RTP as a powder, is then mixed with alcohol and water, filled in vials, labeled and packaged. All of the employees who will work on the line making Havalen will have to wear special clothing - from scrubs, hairnet, booties and gloves to two layers including a whole-body suit - for protection and to ensure the liquid isn’t contaminated.
Havalen faces competition from two other recently approved treatments for advanced breast cancer, but Eisai projects Havalen will become a blockbuster seller, generating about $1 billion per year. Up to 40 production workers could crank out as many as 4 million vials of Havalen on the RTP plant’s commercial production line per shift, Errico said. A second shift could be added.
A second production line is reserved for smaller batches of injectables used in clinical trials.
Errico estimated that initial demand for Halaven will keep about 25 percent of a shift busy. About 30 people have been hired and trained to work in the new plant. Errico said Eisai is looking for contracts to make other injectible products. “Our goal is to be a multi-product facility,” he said.
A framed architectural drawing on the wall shows three production lines Eisai could add on the southside of the building. But those are long-term plans. “I’ll probably be retired before we fill that,”Errico said.